Government mail service may be affected by the Canada Post labour disruption. Learn about how critical government mail will be handled.
Overview
Successful crop production requires that crop pests and diseases be managed so that their effects on the plants are minimized. The management of crop diseases is directed at preventing the establishment of diseases and minimizing the development and spread of any diseases that become established in the crop. Managing pest problems is directed at preventing pest populations from becoming too large and uncontrollable (Portree 1996). The presence of pests and diseases are a fact of crop production and growers must use all available options and strategies to avoid serious pest and disease problems.
Integrated pest management (IPM) is a term used to describe an evolving process where cultural, biological, and chemical controls are included in a holistic approach of pest and disease control (Howard et al 1994). Key components of effective pest and disease control programs include:
- sanitation,
- crop monitoring,
- cultural control,
- resistant cultivars,
- biological control and chemical control
(Howard et al 1994, Portree 1996)
Sanitation - greenhouse clean-up
Greenhouses are completely cleaned and disinfested at the end of each production year. This operation sets the stage for the new crop by ensuring that there are no pest and disease problems carrying over from the previous year. The old crop along with the growing media is completely removed from the greenhouse.
The interior greenhouse structure is pressure washed with a detergent solution and then rinsed. A wash with a 10% bleach solution followed by another rinsing can also be used to ensure that any disease organisms or pests are disinfested from the structure. Do not combine the detergent with the bleach in a one wash operation this avoids chemical reaction between the detergent and bleach. Wear protective gear, including a respirator, when pressure washing the greenhouse to avoid prolonged direct exposure to the bleach solution.
All dripper stakes, clips and truss supports should be soaked overnight in a 10% bleach solution and then rinsed. Irrigation lines should be flushed with nitric or phosphoric acid at a pH of 1.6 to 1.7 (1 part acid to 50 parts water) (Portree 1996). Do not allow the pH or E.C. sensors to be contacted by bleach or acid as they can be damaged (Portree 1996). The acid solution should be held in the irrigation lines for 24 hours before the lines are thoroughly rinsed with water.
CAUTION: Always provide good ventilation through the greenhouse when flushing the lines with acid to avoid the build-up of fumes. Test the pH of the flushed water ensure that the flushed water does not have a pH of below 5 as dangerous chlorine gas may form (Portree 1996).
New sawdust bags should be used for every crop, rockwool slabs can be reused for two or three years (Portree 1996). Slabs that have lost more than 10% of their original height should not be reused as the structure will have changed substantially and will result in yield reduction (Portree 1996).
Steam sterilization of rockwool slabs is a prerequisite to reuse, the slabs should be heated to 100°C and held at this temperature for 10 minutes or held at 75°C for 20 minutes (Portree 1996). Rockwool slabs that have been stacked on palettes and wrapped in heavy plastic require 5 hours to reach 100°C.(Portree 1996).
Crop monitoring
Crop monitoring is the continually on-going surveillance to detect the presence of a pest or disease at the very early stages of development of the disease or pest population, before economic damage has occurred (Howard et al 1994). Everyone involved in working the crop should be made aware of the common pest and disease problems and what to look for to detect the presence of problems in the crop. In addition this general surveillance of the crop, dedicated monitoring of the crop should be included in the weekly work schedule (Howard et al 1994, Portree 1996).
Blue sticky cards, placed throughout the crop, are a useful monitoring tool to help trap and detect pest problems before they become a problem (Howard et al 1994). Yellow sticky cards are known to attract and catch some biological control agents, for example, Aphidius sp. (Don Elliott, pers comm). Biological control agents can be released well in advance of any pest population explosion thus allowing for the establishment of the control agents and prevention of a serious pest problem.
Crop monitoring should begin when the crop is still on the seedling table or at the transplant stage (especially when transplants are obtained by another greenhouse ie. purchased from a propagator) (Portree 1996). If transplants are being purchased from a propagator it is important to be in contact with the propagator regarding any pest problems encountered during the production of the transplants.
It is also important to know what pest control measures were used, if any. It is advisable to establish in advance, what control measures you are willing to have applied to the transplants at the propagator's prior to receiving them into the greenhouse. The concern is that any pesticides that are applied are compatible with the pest control programs i.e. biological control programs that will be used for the duration of the crop. Some growers may insist that only biological controls be used during the production of transplants, and/or that biological control agents be introduced preventively to the transplants before they are received.
Cultural control
Cultural control involves providing the conditions that favour the growth, development and health of the crop, and where ever possible, providing conditions that work against pest and disease (Howard 1994, Portree 1996). Many disease causing fungi and bacteria require the presence of free water or condensation on the plants in order to cause disease (Jarvis 1992, Howard 1994, Portree 1996). High relative humidity promotes the development of disease, and maintaining the environment below 85% relative humidity will help to escape or avoid disease problems (Jarvis 1992, Howard 1994, Portree 1996). Ensuring proper ventilation and air movement within the crop canopy, as well as maintaining optimum plant spacing and a relatively open canopy, will ensure good air circulation and minimize the establishment of micro-climates that favour disease development. Proper contouring of the greenhouse floor will avoid the pooling of water which contributes to localized high relative humidity.
Optimizing the greenhouse environment to favour the development of the plant will ensure a strong, healthy plant which is not only a prerequisite for high yields but also results in plants that are better able to resist diseases and insect pests (Jarvis 1992, Howard 1994, Portree 1996).
Good crop sanitation is another important component of successful cultural control. The plants must be pruned and maintained on schedule, all crop debris should be promptly removed from the vicinity of the greenhouse. Any weeds that happen to gain a foot hold through gaps in the floor plastic should be removed immediately upon discovery and the floor repaired. Personal plants or "pet" plants should not be grown in the greenhouse. Both weeds and "pet" plants can be as source and "haven" for pest and disease problems.
Pruning tools and other equipment should be cleaned and disinfected on a regular basis. Aprons or other clothing worn by the workers should be washed frequently. When a disease or pest problem area exists in the greenhouse, that area of the greenhouse should be worked last, to avoid the spread of the disease or pests by the workers. In this situation, special care must be taken to disinfect tools and to clean clothing.
Maintain a 6 to 10 meter wide buffer zone (Howard et al 1994, Portree 1996) around the outside of the greenhouse by regularly mowing any weeds that try to grow in this zone. The presence of plants in close proximity to the greenhouse can serve as a reservoirs for continual introduction of pests and diseases into the greenhouse. Screening of the intake vents can also play an important role in excluding pests from the greenhouse. It is not enough just to screen-off the intake vents as the screening restricts the air flow into the greenhouse, it is important to ensure that the surface area of the screening used is large enough so that it does not restrict the flow of air into the intake vents (Chang 1996). This may require that a screen 'chamber' be constructed over the vent.
Resistant cultivars
Plant breeders have had considerable success in developing cultivars that contain genetic resistance or tolerance to diseases. When selecting the cultivars to be grown, it is important to consider the genetic resistance of the cultivars to the prevalent disease problems in the region (Howard et al 1994).
The development of cultivars possessing genetic resistance to pests has been relatively unsuccessful (Howard et al 1994), however, the techniques of genetic engineering have made inroads in conferring pest resistance in plants. Genetically modified, pest resistant plants may become available to greenhouse growers in future. The development use of genetically modified plants or genetically modified organisms (GMOs) is currently a contentious issue, and may not be accepted by growers or consumers.
Biological control
Biological control uses beneficial organisms, primarily predators and parasites, to control pest populations below economically important levels. The goal is to establish a balance between the pest population and its parasites and predators to keep the pest population under control. The complete eradication of the pest population is not the goal of biological control programs, as some pest organisms are required so the parasites and predators can reproduce.
The greenhouse industry has a well established reputation for using biological pest control agents more than any other crop production industry. The reason for this is, in part, due to the ability of greenhouse growers to manage the environment to favour the biological control agents. Another factor is the relatively limited number of pest species in greenhouses, as well as a general tolerance of greenhouse crops to leaf damage caused by these pests. The high value of greenhouse produce is another reason why the use of biological controls is economical in greenhouse crops. The increased use of biological controls has led to a reduction in pesticide applications as the industry leads in environmentally responsible, intensive crop production.
Effective biological control of diseases is a more difficult goal and to-date, has rarely been achieved (Howard et al 1994). However, research in developing biological controls for greenhouse crop diseases is ongoing and it is likely that biological control products for greenhouse diseases will be available in Canada in the near future. The primary strategy of biological control for greenhouse plant diseases is to introduce fungal parasites to control populations of disease causing fungi in the greenhouse environment so that they are unable, or have a reduced ability to infect the plants. Some of the promising biological control agents, for example, fungi in the Genus Trichoderma are strong competitors of the disease causing fungi such as Botrytis cinerea, and can be used to protect wound sites to prevent Botrytis from colonizing the wounds.
Chemical control
Pesticides are valuable tools when used as a component of an integrated pest management program (Howard et al 1994). Insecticides should be applied only in support of biological control programs, dealing with localized pest outbreaks in the crop that have escaped the biological control agents. When insecticides are used, care must be taken to ensure that they are compatible with biological control agents, that there will be minimal long term adverse residual effects on biological control programs. Fungicides are used only when a disease problem is detected.
Pesticides are regarded as the controls of last resort because their misuses creates high-profile environmental and food safety problems (Howard et al 1994). Also, the application of some pesticides to a crop can cause stresses that reduce the productive life of the crop and can make the plants susceptible to other pests and diseases (Howard et al 1994). If the use of biological control agents is to obtain a balance between pests and predators that does not threaten the productive yield of the crop, the indiscriminate use of pesticides creates imbalance and uncertainty in the crop. A list of pesticides registered for use on greenhouse peppers in Canada is included in Appendix
Common physiological disorders of tomato

Figure 1. Blossom end rot.
Blossom end rot
Blossom end rot is the most common physiological disorder of greenhouse tomato in Alberta. The disorder is due to a calcium deficiency in the fruit and can occur even when there are acceptable calcium levels in the fertilizer feed solution.
Calcium is an immobile nutrient and once the calcium has been placed at a specific site within the plant the calcium cannot be redirected to another site that may be lacking.
The transpiration process is the mechanism by which nutrients, including calcium, are brought up from the roots and distributed to the developing organs of the plant, including the fruit. As water is lost from the leaves during transpiration there is a natural pull from the leaves to draw more water and nutrients to them. Fruit, on the other hand, do not transpire, or transpire at very low levels due to the lack of stomata. This requires that calcium and the other nutrients necessary for proper fruit development, be directed to the fruit.
Any condition that stresses the plant and interrupts the flow of calcium to the fruit can result in localized calcium deficiencies in the fruit and the subsequent development of blossom end rot.
The most obvious symptom of blossom end rot is the development of a dry, sunken, dark brown or black area at the blossom end of the tomato (Howard 1994, Portree 1996). The symptoms of Blossom end rot can also be completely contained within the fruit (Howard 1994), in which case they are recognized by the blackening of internal tissues usually within the bottom quarter of the tomato fruit.
Conditions that can result in blossom end rot include fluctuating levels of fertilizer feed to the plants or other factors that induce water stress on the plant such as inadequate water/fertilizer feed to the plants (Portree 1996). Fluctuating light levels are also associated with the development of blossom end rot primarily through the resulting fluctuation in uptake of water and nutrients from the fertilizer feed.
High levels of competing cation nutrients in the feed, specifically potassium and to a lesser extent, magnesium can interfere with and restrict calcium uptake. Potassium is the nutrient most often involved in restricting calcium uptake because increasing potassium levels in the fertilizer feed is employed as a strategy to correct blotchy ripening another physiological disorder that affects tomato.
Prevention is the best approach to controlling blossom end rot. Always ensure that there are adequate levels of calcium in the feed and that the plants are receiving adequate fertilizer feed. Maintain overdrain targets at 25 to 30% during the high light periods and pay particular attention to the delivery of water during periods of fluctuating light levels. In this situation care must be taken to ensure that the plants are not overwatered during instances of low light and are not stressed by periods of underwatering when light levels rapidly increase. Ensure that optimum vapour pressure targets (VPD) are met (a discussion of VPD is included in "Commercial Greenhouse Production in Alberta").
Always be aware of the natural cycle of light levels during the year and anticipate the increased water requirements by the plants. For example, the light levels in Alberta increase dramatically in late May and June. Failure to increase the volume of the fertilizer feed delivered will result in water stress which in turn can bring on Blossom end rot.
Pay close attention to the daily per cent of overdrain as this will be the first indicator of the need for increased fertilizer feed as the per cent overdrain drops below the optimum 25 to 30%. Daily monitoring of the E.C. also gives clues to the relative water stress on the plants.
High E.C., above 3.5 mmho, can place a salt-induced water stress on the plants and result in the onset of BER. Maintain the feed E.C. to ensure the resulting rootzone E.C. is within acceptable limits for the cultivar you are growing. The acceptable level for most cultivars, under high light conditions, is usually in the vicinity of 3.5 mmho. When considering dropping the feed E.C. always ensure that the volume of water delivered to the plants is optimized first. Any adjustments to the E.C. of the feed solution should be made gradually, in 0.1 mmho increments per day.
Remember that the plants on the outside rows of the greenhouse will require more water that the plants in the interior of the crop because they receive more light. This is especially true of the plants on the east, south and west sides of the greenhouse. These plants may require additional drippers to maintain adequate overdrain.
The strategy of increasing the drippers from 1 to 1.5 or 2 drippers per plant (2, 3 or 4 drippers per bag) allows for more water to go to these plants without increasing the volume of water delivered to the majority of the plants beyond the 25 to 30% overdrain.
Blotchy ripening
Uneven colouration of the ripening fruit is symptomatic of this disorder (Howard 1994, Portree 1996). Blotchy ripening is primarily considered to be a physiological disorder but can also be a result of virus infection (Howard 1994, Portree 1996). A number of conditions can bring about blotchy ripening of the fruit including: inadequate fertilizer levels, high fruit temperatures, and infection by tomato mosaic virus.

Figure 2. Mature tomato fruit exhibiting blotchy ripening.
When fruit temperatures exceed 39°C the red pigments of the fruit can be destroyed, the less heat sensitive yellow pigments remain. The overall effect is the blotchy yellow and red fruit. If the blotchy ripening is occurring primarily on fruit that is exposed to the direct sunlight, the cause of the problem is probably high fruit temperature. Be aware that air temperature is not a good indication of fruit temperatures which can rise to over 10°C above air temperatures. Infrared thermometers can be used to measure the temperature of an object and will give an accurate reading of fruit temperature.
Plants in the outside rows are the first to be affected by temperature induced blotch ripening since they have the most exposure to the direct sunlight. Applying shading to the greenhouse is standard practice in Alberta greenhouses, particularly in the south, and will help to reduce the occurrence of blotchy ripening. Adjusting pruning practices to leave more leaf cover to reduce fruit exposure to direct sunlight will also have a dramatic effect in reducing incidence of blotchy ripening.
Remember plants that are out of balance can exhibit blotchy ripening. Overly generative will have smaller leaves and may not be able to provide the required canopy to protect the fruit. Plants that are overly generative during the high light season will only tend to become even more generative overtime and yield can be severely reduced. Also ensure that the plants are twinned well in advance of the high light season to ensure the summer planting density targets are met which will, in turn, provide an adequate crop canopy.
Plants that are overly vegetative can also exhibit blotchy ripening through directing too much of its resources towards the production of leaves and stems at the expensive of the developing fruit. Fruit to leaf ratios of 20 - 23 fruit to 14 - 15 leaves represent optimum targets and result in reduced likelihood of blotchy ripening (Portree 1996).
Inadequate nutrition to the plant can also result in blotchy ripening.
Daily monitoring of the feed and leachate E.C. will ensure that the overall level of fertilizers has not drastically fallen off because of an equipment malfunction. In situations where the fertilizer feed and the leachate E.C.s are optimum, and blotchy ripening is occurring, attention is then focused on the fertilizer feed targets and a nutrient analysis is conducted.
Excessive nitrogen can also play a role in the occurrence of blotchy ripening (Howard 1994). Nitrogen favours vegetative growth and an overly vegetative plant results in inadequate resources being directed to the fruit. Inadequate levels of potassium can also result in blotchy ripening. A feed analysis will confirm the levels of nutrients in the fertilizer feed. It is important to ensure that the fertilizer feed targets are being met before any other adjustments to the feed are considered.
In the situation where the nutrient feed targets are being met, all other reasons for blotchy ripening have been ruled out, attention turns to the levels of potassium in the feed. In most situations where increasing the potassium levels is considered, the actual potassium levels in the feed have not fallen below the levels initially targeted.
Increases in the level of potassium in the feed should be done in increments of 10% every two weeks to a final maximum 20% increase in potassium. The concern with increasing potassium is related to the potential interaction with calcium uptake. Both potassium and calcium are positively charged ions, or cations, and can be considered as being in competition with respect to access by the plant. The main point; too much potassium can interfere with calcium uptake and cause a blossom end rot problem.
When increasing potassium levels in the feed first ensure that the environment and amount of fertilizer feed delivered is optimized with respect to calcium uptake. If these conditions are met and blotchy ripening still persists, increasing the potassium levels should correct the problem. If further increases in potassium are contemplated, ensure that calcium is also increased at the same relative rate as not to change the calcium/potassium ratio any further.
Blotchy ripening can also be aggravated by tomato mosaic virus infection (Howard 1994, Portree 1996). Using tomato cultivars that are resistant to tomato mosaic virus (TMV) will help avoid TMV associated blotchy ripening. Practically all new greenhouse tomato cultivars have adequate TMV resistance; TMV is more of a concern on older cultivars.
Cuticle cracking or russeting
This disorder is identified by the presence of fine cracks at the top of the fruit which reduces the appearance and quality of the fruit and becomes more noticeable as the fruit ripens (Portree 1996). The fruit on plants that are out of balance, outside the optimum the fruit to leaf ratio, and either too vegetative or too generative, have a greater tendency to exhibit cuticle cracking.
Overly vegetative plants tend to have larger, fewer fruit that develop more slowly and have a tendency to exhibit cuticle cracking (Portree 1996). Overly generative plants have more fruit exposed to direct sunlight, higher fruit temperatures because of reduced leaf lengths and overall canopy cover and a tendency for cuticle cracking. The disorder is also more likely under conditions of high humidity (Howard 1994), over 85% and when temperatures are too cool during fruit set (Portree 1996). Higher root zone E.C. generally results in less cuticle cracking.
Cuticle cracking is also associated with late day watering applications. As the night period begins and the natural light falls off with the sunset, the greenhouse air temperatures drop to meet the night time temperature targets. As the air temperatures and light levels drop, the plants also begin to cool and transpiration is reduced. The rootzone, however, can still be quite warm and the roots still active with the uptake of water and nutrients continuing for some time. With the drop in transpiration, less of this water is drawn to the leaves and more is directed towards the fruit. This "burst" of nutrients directed to the fruit can cause a relatively rapid expansion of the fruit that subsequently results in cuticle cracking.
Cuticle cracking can be avoided by maintaining optimum growing conditions and managing the fruit load and leaf number to ensure optimum plant balance. If cuticle cracking begins to occur on well balanced plants in an optimized environment, the last watering event of the day may be occurring too late. Moving the last watering event back by one half hour should help to correct the problem. Always ensure that adequate water is applied over the course of the day to meet overdrain targets.
Low temperatures during flowering – Catfacing
Catfacing is and example of a fruit disorder caused by low temperatures during flowering. Air temperatures that are below the optimal temperatures for pollination can result in fruit deformity.

Figure 3. Catfacing.
Fruit that are affected by catfacing are misshapen with black scarring of the blossom end. If problems with misshapen fruit begin to occur in the tomato crop the first step in correcting the problem is a thorough review of actual air temperatures at the flowers.
Magnesium deficiency
This disorder can occur in the leaves of vigorously growing, high yielding crops even when magnesium levels in the fertilizer feed are optimum. Symptoms begin as light green spots between the veins of the leaves, with the spots becoming yellow and eventually purple in colour as the symptoms intensify. The symptoms are first noted on midway down the plant, about twelve leaves down from the top.

Figure 4. Magnesium deficiency in tomato plant.
The development of magnesium deficiency symptoms is dependent on cultivar. Affected crops do not generally exhibit yield loss. Foliar application of magnesium fertilizer does not alleviate the symptoms. Growers who are unfamiliar with this condition will have an analysis of the fertilizer feed solution to confirm that nutrient levels, including magnesium, are adequate.
Truss kinking
Under conditions of low light the flowering truss can elongate resulting in a weakened truss that kinks under the weight of the developing fruit (Portree 1996). The kinking interrupts the flow of nutrients to the fruit and results in slower fruit development and poor fruit size. Truss kinking can be prevented by using truss supports. Truss supports are typically used on the first eight trusses on the plants.

Figure 5. Truss supports are usually placed on the first eight trusses in order to prevent truss kinking.
pH induced leaf yellowing
A rapid onset of leaf yellowing in the lower to middle canopy can occur after a dramatic drop in the pH in the root zone. Symptoms have occurred when the root zone pH had fallen to 3.5 from the ideal 5.8. The cause of the pH drop is usually due to equipment malfunction that results in over dosing of acid into the fertilizer feed. The affected leaves do not recover and the extent of the overall damage to the plant is dependent upon the magnitude and duration of the highly acid conditions.

Figure 6. Leaf yellowing induced by a rapid drop in the pH of the nutrient solution.
Herbicide drift
Herbicides are not recommended for use in greenhouses however greenhouses can be located in agricultural areas and there is the potential for herbicides to enter the greenhouse via the vents. Some growers have used herbicides to control weeds around the greenhouse, this is a risky practice as herbicides can cause considerable crop damage if they enter the greenhouse. The damage caused by herbicide drift depends on the specific herbicide.

Figure 7. 2,4-D injury on the young leaves.
If you are in an agricultural area make sure your neighbours know that you are a tomato grower and that your crop can be seriously damaged by herbicide drift. Make sure that they notify you before they apply a herbicide to their crops so that you can take the necessary measures to close up the greenhouse during the application. They should also be aware to not apply herbicides when the wind is blowing towards the greenhouse. Never use herbicides to control weeds around the outside of the greenhouse, use mechanical methods of weed control.
Fungal diseases of greenhouse tomato
Pythium root rot and wilt (Pythium spp.)
Pythium root rot and wilt can affect tomato plants at any age. Pythium is a very common genus of fungi that are classed as water moulds. For practical purposes it is wise to consider that Pythium is always present in the greenhouse at some level. As with most diseases, the presence of the pathogen does not guarantee that the disease will develop.
Pythium takes advantage of plants under stress and this is especially true for tomato plants. A healthy tomato crop rarely has trouble with Pythium. Avoiding problems with Pythium through maintaining healthy plants in optimum growing environment is the best way to deal with Pythium.
Pythium attacks the roots of the tomato plant and concentrates on the small feeder roots (Portree 1996). In situations where Pythium infected tomato plants have been examined, the bulk of the roots can remain clean and white, suggesting a healthy root system. When sections of these roots are examined in the laboratory they often reveal only very low levels of Pythium, levels far below what would be expected if Pythium was a problem. The feeder roots where Pythium is causing the problem are often too small to work with or overlooked in the examination of the roots of Pythium wilted tomato plants.
The main conditions that predispose roots to infection by Pythium are physical damage to the roots and overwatering. Physical damage is really only a concern during the transplanting stage where the seedlings are placed into the production greenhouse. Care must be taken to minimize the damage that can occur during handling, transporting and transplanting of the tomato seedlings. Overwatering is much more of an ongoing concern with respect to Pythium infection. Overwatering causes the roots to suffer from a lack of oxygen which stresses the plant and predisposes it to infection.
There is a pattern that often occurs in tomato greenhouses where Pythium has become a problem that demonstrates that the problem with Pythium occurs as a result of watering practices over a considerable period of time after transplanting.
Once the tomato seedling has been transplanted into the production greenhouse, the first priority is to establish the plant in the growing media by encouraging the development of a strong root system. A careful watering strategy is employed to deliver enough water to meet the requirements of the plant (indicated by meeting the overdrain targets of 10 to 15%) and to deliver the water usually with one hour intervals between watering events.
The strategy is designed to ensure the plants are not stressed due to lack of water, and provide enough time between watering events to avoid waterlogging and ensure a healthy root environment. Timing watering events every hour encourages the roots to grow and actively seek water compared to more frequent watering events which are constantly delivering water to the young roots. The concept is that more frequent watering events encourages a less well developed root system.
Unless there so much water applied that the root systems are actually subjected to waterlogged conditions, Pythium rarely develops in the seedlings at this stage. The problem occurs as the light levels increase and the transpiration stress is increased requiring the root system to supply the increased water requirements of the plants. The problem usually manifests itself during the onset of the high light period which occurs at the end of May or early June.
Plants that have an inadequately developed or inadequately functioning root system cannot meet the light induced increased demand for water. The result can be dramatic with the entire crop wilting. Growers often react to this by applying more water, using the logic that a wilting crop is not getting enough water. The problem at this point is with the root system, not the amount of water, and applying more water can waterlog the rootzone further stressing the plants, and encouraging the development of Pythium.
Growers who hold the course on watering by using the percentage overdrain as their guide, will generally see a recovery of the plants within about 2 weeks of the high light induced wilt. Growers who apply excessive water will see a continual decline and the onset of problems with Pythium and plant death. In both situations yield will suffer with the most severe losses associated with the prolonged overwatering.
The best way to avoid the high light induced wilt, and the development of Pythium from overwatering, is to always use optimum overdrain targets to determine the amount of water delivered to the plant. Daily monitoring of the overdrain levels is a very important tool for making decisions concerning changing the amount of water delivered to the plant. Increase the amount of water when the overdrain falls below the 10 to 15% target. Monitoring the E.C. of the overdrain provides the basis for adjusting the overdrain target.
For most tomato varieties, the E.C. in the rootzone should be kept in the range of 3.5 to 3.7 mmho. As the light levels increase the E.C. in the rootzone will increase as the plants have a higher relative requirement for water over nutrients and the fertilizer salts can accumulate. Increase the overdrain targets as the E.C. rises past 3.7 mmho to a maximum of 25 to 30%. If the rootzone E.C. continues to increase beyond 3.7 mmho at 25 - 30% overdrain, decrease the feed E.C. 0.2 mmho every 2 to 3 days until the rootzone E.C. stabilizes at 3.5 mmho.
Mature producing tomato plants under high light summer conditions will use up to 2.5 litres of fertilizer per day with overdrain targets of 25 to 30%. If wilting occurs under these watering conditions and water is increased to alleviate the wilting, the per cent overdrain will also increase beyond 40% indicating that the plant is not using the extra water. The problem with wilting will not be corrected with the application of more water, because the base cause of the wilting is an inadequate root system, not insufficient watering. The addition of extra water increases the problem by fostering conditions that further the development of pythium root rot.
In short, conditions that increase stress on the root system can provide an environment that is favourable to the development of pythium root rot. Optimized watering practices will prepare the plant for the high light conditions and will minimize the waterlogging conditions which foster Pythium infection.
Other management practices to prevent Pythium and root diseases in general include; avoiding high water temperatures (temperatures over 24oC), controlling fungus gnats and shore flies (which spread the Pythium spores from plant to plant), careful removal and destruction of infected plants, avoiding introduction of soil to the watering system and using new sawdust bags for every crop. Rockwool slabs should be sterilized between every crop.
Fusarium Wilt (Fusarium oxysporum f. sp. lycoperisici)
Fusarium wilt usually occurs when the tomato plants are under the stress of full fruit production and high summer temperatures (over 28°C). The problem occurs more commonly in greenhouses that are not equipped with evaporative cooling systems. Other factors that have been associated with the onset of this disease include any shock to the root system from irrigation system malfunctions causing either a lack of water during a few hours, loss of pH control resulting in low rootzone pH (pH of 3) and high rootzone E.C. (greater than 5.0 mmho). The risk of fusarium wilt is always increased when these predisposing problems occur during hot days.
In any situation where fusarium wilt occurs, the pathogen has to be present. Predisposing problems only set the stage for aggressive fusarium infection of the root system.
The onset of symptoms begins with wilting and yellowing of the lower leaves (Howard 1994, Portree 1996). The symptoms progress up the plant as more leaves are affected. It is not unusual for wilting and yellowing to occur only on leaves from one side of the stem, with leaves on the opposite side of the stem seemingly normal (Howard 1994, Portree 1996). The yellowing is commonly a more distinctive orange-yellow colouration on affected leaves that can be diagnostic of fusarium wilt. Infected plants eventually completely wilt and die.
The fusarium wilt fungus is introduced into the root system of plants via the seed or soil (Howard 1994, Portree 1996) and plants can remain symptomless for quite some time and the disease may only develop when the plants come under stress.
Control through prevention, using fusarium wilt resistant cultivars, using new growing media for each crop, and maintain optimum growing conditions. Infected plants must be carefully removed and destroyed.
Fusarium crown and root rot (Fusarium oxysporum f.sp. radicis-lycopersici)
This Fusarium disease is caused by a close relative of the fusarium wilt fungus. Symptoms of fusarium crown and root rot first appear as wilting of the upper leaves, usually developing at the onset of the high light period when the plants are begin full fruit production ( 1994, Portree 1996). Examination of the base of the stems will reveal a dark brown stem canker. Some development of adventitious may be apparent above the stem lesion (Howard 1994). A reddish discolouration is present in the vascular tissue which can extend 25 or 30 cm up the stem from the base. The roots are typically brown and discoloured.

Figure 8. Typical orange chlorosis of plants infected by Fusarium oxysporum f. sp. lycopersici.
Infected plants must be carefully removed and destroyed. Do not reuse growing media from infected crops. Do a thorough clean-up of the structure between crops. Ensure that the entire greenhouse floor is securely covered in plastic and that there is no exposed soil. The fungus survives well in soil and can carry over one year to the next and can enter the greenhouse through holes in the plastic floor. Use resistant cultivars or tomato plants which have been grafted onto resistant rootstocks (Portree 1996).
Botrytis blight (Gray mold) (Botrytis cinerea)
Botrytis cinerea has a worldwide distribution and causes serious losses to a wide range of greenhouse crops and field crops (Howard et al 1994, Hausbeck and Moorman 1996) Botrytis is truly a disease of opportunity, it is considered one of the easiest diseases to control through proper management of the environment (Jarvis 1992, Howard et al 1994).
Conditions of high relative humidity, over 85%, and the presence of water droplets on the plants favour disease development. All above "ground" parts of the plant can be affected; leaves, petioles, stems and fruit (Howard et al 1994, Portree 1996). Botrytis is quick to colonize wounds on the plant.
The symptoms first appear as a dark discolouration in the plant tissue around the wound. As the disease progresses, more tissue is affected and the lesion becomes larger and a gray mass of spores can develop as the fungus grows on the surface of the lesion producing a velvety mat over the lesion. As conditions change and the relative humidity drops for a period of time, the gray spore mass can disappear leaving a tan coloured lesion. If the relative humidity around the lesion increases again, the fungus can again begin to grow out onto the surface of the lesion.
The most severe symptom associated with botrytis blight is the stem canker. Plants can be girdled and killed by these cankers. Through the routine handling of the crop, pruning and harvesting, wound sites suitable for infection are created almost daily. If a Botrytis infection starts at a petiole stub where a leaf has been removed, the infection can move into the main stem and girdle the stem.

Figure 9. Botrytis stem canker with a mass of gray spores that have formed under conditions of high humidity.
Tomato fruit can also be affected and can exhibit a peculiar symptom referred to as ghost spots. The symptom is most prominent on ripe fruit but can be first noticed at the mature green stage (Howard et al 1994, Portree 1996). The ghost spot symptom appears as a light coloured halo around a small necrotic spot on the tomato fruit (Howard et al 1994, Portree 1996). The necrotic spot in the middle of the ghost spot is caused by the aborted infection of the fruit where a Botrytis spore has landed. The presence of free water for a period of 4 to 6 hours (Portree 1996) is necessary for Botrytis spores to germinate and to begin to infect the plant.

Figure 10. The ghost spot symptom on tomato fruit.
The presence of free water on the fruit allows the spores to germinate and begin to infect the fruit. The failure of the infection to actual take hold is associated with the length of time that the free water is actually present. The free water being present long enough for the infection to begin, but not present long enough to ensure that the infection can continue.
Fruit can also become infected at the calyx where free water is likely to remain longer in the depression at the calyx end of the fruit which can hold water much like a small basin (Howard et al 1994). The light halo around the small necrotic spot is due to the physiological response of the fruit to the initial infection and is associated with the defence response of the plant.
The presence of ghost spot symptoms on the fruit results in downgrading of the affected tomatoes.
The most effective approach to control is to manage the environment to avoid the formation of free water on the plants. Before the environment can be effectively managed to avoid conditions that are favourable to the development of Botrytis it is useful to know the types of conditions that result in the formation of free water on the plants.
Botrytis is a problem in tomato crops in the spring and fall. There are some typical situations that lead to the formation of free water on the plant, all of which center around condensation. Warm air can hold more water vapour than cold air. The basic requirement for condensation is a warm, humid air mass contacting a cold surface. As the warm air is cooled by contact with the cold surface, the air loses some of the water vapour to the cold surface. The result is the vapour condenses directly on the cold surface.
When warm, humid greenhouse air contacts cold surfaces of the roof, there can be formation of condensation on the roof structure. The condensation can then drip directly onto the plants. The second situation is associated with conditions that cause the air temperature to warm faster than the plant temperature which allows for the condensation of water from the air directly onto the plant surfaces.
This usually occurs early in the morning when air temperatures ramp more quickly from the night temperature to the day temperature than does the temperature of the plants themselves. As the air temperatures ramp, and the plants begin to transpire, the water vapour in the air increases. The relative humidity increases and if the greenhouse is not aggressively dehumidified, condensation forms on the cooler plant surfaces. As the morning progresses, the plant temperatures increase to where they match or exceed the air temperature and condensation onto the plant surfaces stops.
This second situation typically occurs in late spring, when outside temperatures are still below freezing, and the natural light levels are good. The greenhouse air mass can warm quite quickly after sunrise due to solar gain. As the air temperature rises, and the plants begin to transpire due to the incoming light, the relative humidity of the greenhouse air increases. As the temperature and humidity of the air increases there will be a point where condensation forms on the plants.
The primary approach to avoiding condensation onto the greenhouse structure and plants is through dehumidification. Dehumidification is accomplished through heating and venting. The warm, moisture laden greenhouse air is exhausted when the relative humidity reaches 75 - 80%. As the warm humid air exits the greenhouse, colder, drier air is drawn in the vents. This air has to warmed and mixed with the greenhouse air mass to avoid cold air contacting the plants and causing chilling injury.
Simply put, dehumidification maintains optimum relative humidity and temperature targets in the greenhouse where the air mass is constantly taking up more water vapour due to the active plant transpiration. Dehumidification ensures conditions for optimum plant transpiration, which in-turn ensures optimum yield. Dehumidification also ensures that condensation does not develop directly on the plant surfaces, or that excessive condensation forms on the structure that subsequently rains down on the plants.
In the case where condensation forms on the plants early in the morning because the plant temperature lags behind the air temperature as it is warmed by the early morning sun, it is advisable to begin to heat the greenhouse well so that the plants are warmed to the daytime temperature target before sunrise.
In Alberta, the ghost spot symptom of Botrytis occurs when condensation forms on the plants due to cold plants in a warm air mass during the early morning hours. Ensuring that the plant temperature is at the daytime target in advance of sunset effectively eliminates the symptom.
Effective dehumidification ensures that the formation of condensation on the greenhouse roof and subsequent dripping onto the plants is minimized, and minimizes the opportunity for Botrytis to infect these wet sites on the plants. More effort to protect plants from dripping from problem drip sites is often required to completely deal with the problem.
Botrytis will usually establish a foothold in the greenhouse early in the season. As the season progresses into the summer, the problem practically corrects itself by virtue of the warm dry Alberta summer air. With the onset of fall, the conditions change to favour the build-up of humidity in the crop as the outside air temperatures drop. In most cases when there is a problem with Botrytis in fall, there was almost certainly a problem with it in the spring. It is only rare instances when Botrytis becomes a problem in greenhouses only in the fall, with no problem having occurred in the spring.
Early and late season problems with Botrytis are almost always associated with poor dehumidification strategies. Effective dehumidification is aggressive dehumidification to ensure that the relative humidity in the greenhouse does not exceed 80% and that the plant temperatures are always within about 5°C of air temperatures.
Dehumidification requires the simultaneous heating and venting of the greenhouse, which requires the input of energy and is results in higher energy consumption and cost. Some strategies to minimize energy costs include marginal approaches to dehumidification, with a resulting high humidity and conditions that favour condensation. Such strategies not only compromise maximum crop production through reduced transpiration and subsequent plant growth, but also encourage the development of Botrytis and subsequent production losses due to disease.
Other strategies to minimize the occurrence of Botrytis include maintaining good air circulation through the canopy, avoiding the pooling of water on the greenhouse floor, ensuring the floor is free of pruned leaves, ensuring that tomato stems do not contact the damp floor. The use of pipe rail heating systems ensure that the floor and canopy remains relatively dry as well as driving the humid air out of the canopy helping to eliminate high humidity microclimates that favour Botrytis. Pipe and rail heating also allows for the maintenance of relatively warm plant temperatures and practically eliminates condensation on the plants.
Powdery Mildew (Erysiphe sp.)
Powdery mildew of tomato was first detected in Alberta greenhouses in 1997. Powdery mildew is common on a number of plants, however the causal organism is not the same for all plants. For example, the powdery mildew on greenhouse tomato is not caused by the same fungus that causes powdery mildew on greenhouse cucumber.
Powdery mildew on tomato appears as diffuse whitish powdery patches on the surface of the leaves, looking much like talcum powder dusted on the leaf. The white powdery material is the fungal mycelium growing on the leaf surface. The symptom is not nearly as distinct as the powdery mildew on cucumber which causes pronounced whitish patches. As the disease progresses, the leaf tissue under the whitish patches begins to yellow.

Figure 11. Powdery mildew on tomato.
The disease usually establishes under conditions of low relative humidity. Plants that are close to furnaces and that are stressed are usually the first to develop symptoms. The disease can spread quickly throughout the crop.
Avoid air temperatures above 21°C, ensure a relative humidity of at least 70%. Overhead misting can be effective in slowing the development of the disease in the early stages of infection (Portree 1996). Chemical controls are available.
Bacterial diseases of greenhouse tomato
Pith Necrosis (Pseudomonas corrugata)
This disease usually occurs in mid to late spring, once the plants have set and begun to develop fruit to mature size. The disease typically strikes in crops where the plants are growing vigorously and have developed thick canopies (Howard et al 1994). The appearance of this disease appears to be counter intuitive, striking plants that appear to be in excellent condition which have been given optimum growing conditions that may have established a slightly over vegetative plant balance.

Figure 12. A blackened pruning scar indicating pith necrosis.
Black lesions develop on the stem and the lesions expand along the length of the stem as the disease progresses. Affected plants eventually wilt and may display yellowing of the lower leaves. Affected stems can eventually collapse at the lesion sites (Howard et al 1994). Examination of the stem interior reveals a dark brown to black discolouration with a ladder-like appearance of the stem due to cross strands remaining intact (Howard et al 1994).

Figure 13. Stem cross section revealing the extent of pith necrosis.
The bacterium is considered to be soil and water borne, but little is understood about its biology (Howard et al 1994, Portree 1996). The disease does not readily spread throughout the crop. Not all affected plants will die. As the plants become more generative and light levels increase, problems with the disease stop.
Severely infected, wilted plants should be carefully removed and destroyed. Ensure that an active growing environment is maintained and that plants are not overly vegetative.
Bacterial canker (Clavibacter michiganensis subsp. michniganensis)
This disease is most common on tomato crops grown in soil. Wilting of the lower leaves is usually the first symptom (Howard et al 1994, Portree 1996). There is an associated light brown streaking along the length of the stem and leaf petioles as the disease progresses (Howard 1994). The affected stems can take on a watery appearance and the streaks can split open to form a canker (Howard 1994). A clear to whitish coloured ooze may be associated with the cankers or wound sites along the length of the brown streaks on the stem.

Figure 14. Black streaking of the stem and bacterial ooze, characteristic symptoms of bacterial canker.
The disease is favoured by conditions of high humidity and is readily spread from plant to plant by splashing water, by infested pruning tools and by the day to day handling of the crop by workers (Howard 1994).
The bacterium can survive in the soil, in infected crop residue from season to season. The bacterium is seed borne (Howard et al 1994, Portree 1996).
Use disease free heat treated seed. Disease plants and adjacent plants should be removed as soon as an infection is discovered (Howard et al 1994, Portree 1996). Control the relative humidity to avoid the formation of free-water on the plants. Do not work the crop by moving from infected areas to healthy areas. Workers should wash their hands after working with infected plants.
Virus diseases of greenhouse tomato
Tomato Mosaic Virus (ToMV)
Tomato mosaic virus is distributed throughout the world and is found wherever tomato is grown (Howard et al 1994, Portree 1996). Tomato mosaic virus has a very wide host range that includes a number of greenhouse crops including: petunia, snapdragon and pepper (Howard et al 1994). In tomato crops the virus is very easily spread from plant to plant during routine handling of the plants (Portree 1996).
ToMV is both seed and soil borne and can survive in the crop residue for up to two years ( Portree 1996). There are a number of strains of the ToMV and the symptoms expressed by the plants can vary with the strain (Howard et al 1994, Portree 1996). Typical symptoms include alternating light green to dark green mottling or mosaic of the leaves and the fern leaf symptom, where the blades of the leaflets undergo a distorted development into a very narrow shape (Howard et al 1994, Portree 1996).
ToMV can also reduce fruit quality when fruit symptoms occur. Infected fruit develop a blotchy appearance which can increase in severity to form sunken areas that are dark brown to black in colour (Howard et al 1994, Portree 1996). Severe infections can cause significant yield losses (Howard et al 1994, Portree 1996).
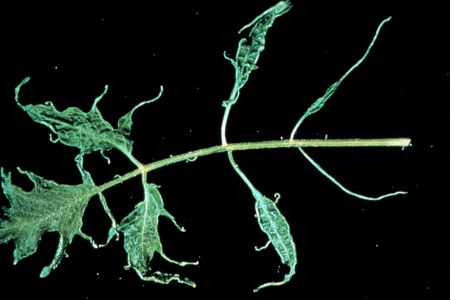
Figure 15. Shoe string leaf symptom of ToMV.
Most modern commercial cultivars contain resistance to ToMV and infected plants rarely develop severe symptoms. The symptoms of ToMV are often more severe under low light conditions, and the fern leaf symptom can develop on some of the resistant cultivars under low light conditions. The infected plants improve and grow out of the symptom as the light levels increase. There is no appreciable yield loss in most cases.
Other control measures include the routine washing of work cloths and disinfection of pruning tools by dipping in a 10% trisodium phosphate solution. Completing a thorough greenhouse cleanup at the end of the crop year, growing in soilless substrates, and ensuring that the soil floors of the greenhouse are completely covered and sealed off with plastic serve to limit the year to year spread of the virus.
Tobacco Mosaic Virus (TMV)
A minor disease of greenhouse crops, TMV has a very wide host range and is one of the most infectious plant viruses (Howard et al 1994). The symptoms of TMV are virtually the same as Tomato mosaic virus (ToMV). Tobacco mosaic is also both soil and seed borne (Howard et al 1994).
Control recommendations for TMV are the same as for ToMV. Tomato cultivars with resistance to ToMV will also have some resistance to TMV (Howard et al 1994, Portree 1996). As TMV can be present in cigarette tobacco it is advised not to smoke near plants or in the greenhouse. Hands should be thoroughly washed with soap and water after using tobacco products to prevent transmission of the virus to the crop (Howard et al 1994, Portree 1996).
Tomato Spotted Wilt Virus (TSWV)
This virus also has a very wide host range infecting a number of greenhouse ornamental and vegetable crops (Howard et al 1994, Portree 1996). As more became known about this virus it was determined that what was thought to be a disease caused by a single virus, was actually caused by two separate viruses. The two viruses are tomato spotted wilt virus and impatiens necrotic spot virus (INSV). Both viruses can infect tomato and cause similar symptoms. If tests are conducted for TSWV they should also include tests for INSV.
TWSV and INSV are not easily spread by handling the plants. The primary vector of these viruses are thrips, particularly the western flower thrips (Howard et al 1994, Portree 1996).
Symptoms of TSWV and INSV include stunting of the plants and bronzing of the leaves (Howard et al 1994). Necrotic lesions can develop on the leaves that progress down the petiole to the stem (Howard et al 1994, Portree 1996). Ring spots of alternating red and yellow can develop on the fruit (Howard et al, 1994). Affected plants can also wilt, examination of the root system will reveal apparently healthy white roots which is not consistent with wilts due to root rot.
There are not TSWV / INSV resistant tomato cultivars. The main approach to controlling this virus is through the control of the thrips vector. The level of thrips control routinely obtained in Alberta greenhouses is usually sufficient so that TSWV and INSV are not a common problem.

Figure 16. Wilting caused by TSWV.
Pests of greenhouse tomatoes and their biological control
Descriptions of the common pests of greenhouse tomatoes are followed with a list of the biocontrol agents recommended for control. Pesticides are not discussed. Pesticide recommendations can be obtained from a greenhouse crop production specialist.
Assessment of the quality of biological control agents
Biological control agents are living organisms and their ability to establish and control pest populations depends on their fitness. When ordering biological control agents ask the supplier what to look for to help assess the quality of the agents when they arrive. A hand lens or magnifying glass is very useful when inspecting packages of biological control agents.
Packages arriving during the winter should be checked immediately to ensure that they have not been frozen or subjected to cold temperatures. The inside of the shipping cooler should not be cold, if the ice pack contained within the cooler is frozen solid, it is likely that the entire package froze and the biological controls have been damaged or killed (Portree 1996). Packages received during the summer months should be cool inside, if they are hot then the biological control agents may be damaged or killed.
Always release the biological control agents into the greenhouse as soon as possible after they arrive. Follow the instructions provided with the package.
Whiteflies
Whiteflies are the most common insect pest of greenhouse tomatoes. There are two whitefly species that are a problem in greenhouse crops; the greenhouse whitefly, Trialeurodes vaporariorum and the silverleaf whitefly Bemisia argentifolii. Of these two, it is the greenhouse whitefly that is almost exclusively found in Alberta tomato crops.
Whitefly adults are approximately 2 millimetre long with the shape of a moth. The adults will fly up from the plants when disturbed, and generally move only a short distance before settling down on another leaf. Adult females lay their eggs on the undersides of leaves which hatch in about 10 days.
Whitefly damage the plant by sucking sap from the leaves. Large infestations can cause leaf yellowing and a general decline in the plant. Sooty mold is commonly found in association with whitefly. As with aphids, whitefly feeding also results in honeydew formation which can reduce fruit quality. The presence of the honeydew and sooty mold can necessitate the fruit be washed prior to going to market. The presence of sooty mold on the leaves can reduce the productivity of the leaf by reducing the amount of light reaching the leaf surface (Howard et al 1994).
The parasitic wasps, Encarsia formosa and Eretmocerus eremicus, are effective against whitefly with parasitized whitefly scale becoming yellow or black in colour, depending on the parasite. Scale parasitized by Encarsia formosa is black in colour. Delphastus pusillus is a small beetle that feeds on whitefly eggs and is ideal for complementing Encarsia and Eretmocerus (Portree 1996).
Thrips
There are two species of thrips that are common pests in greenhouse vegetable crops, the western flower thrips (Frankliniella occindentalis), and the onion thrips (Thrips tabaci) (Howard et al 1994, Portree 1996). Thrips feed by opening wounds on the plant surface and sucking out the contents of the plant cells, the feeding results in small whitish streaks on the leaves and fruit and can cause distortions in the young developing fruit (Howard et al 1994, Portree 1996).

Figure 17. The western flower thrips.
The adult thrips congregate in the flowers and regular monitoring of the flowers will allow for the early detection of thrips. Yellow and/or blue sticky traps placed throughout the crop, as with the other insect pests, will help in the early detection of thrips infestations. Avoid using yellow traps if Apidius sp are being used for the control of aphids in the crop.
In addition to causing direct feeding damage and resultant yield loss, both thrips species are vectors of Tomato Spotted Wilt Virus (TSWV) which can be a serious disease problem in peppers and tomatoes (Howard et al 1994, Portree 1996). One of the main control measures for minimizing the spread and infection of TSWV within the crop is to control the thrips vectors.
There are a number of predators available for biological control of thrips: predatory mites Amblyseius degenerans, Amblyseius cucumeris, Hypoaspis miles and Hypoaspis aculeifer and predatory bugs, Orius insidiosis and other Orius species.
Two-Spotted Spider Mite
The two-spotted spider mite (Tetranychus urticae) is a common pest of a number of greenhouse crops (Howard et al 1994, Portree 1996). Typical symptoms of two-spotted spider mite infestations include speckling of leaves and fine webbing on the underside of affected leaves. As the spider mite population increases, the leaves become brittle and brown in colour, the amount of webbing on the leaves becomes very prominent and the mites can be seen milling about on the webs.
It is very easy for the two-spotted spider mites to be "picked-up" on clothing and transported throughout the crop by workers. As the season progresses into fall, female two-spotted spider mites develop a bright orange-reddish colour as they prepare for the winter. The female mites seek shelter in crevices throughout the greenhouse and a thorough end of season pressure wash clean-up is necessary to minimize the number of females that survive to the next crop.
Biological control of the two-spotted spider mite in tomato can be obtained by introducing the predatory mite Phytoseiulus persimilis as soon as two-spotted spider mites are detected in the crop. The mites Amblyseius fallacis and Amblyseius californicus are closely related to P. persimilis and should be used along with P. persimilis.
Carmine Spider Mite
The carmine spider mite is a variant of the two-spotted spider mite and takes its name from the slight reddish colour that this mite displays. The carmine spider mite distinguishes itself from the two-spotted spider mite through its ability to cause rapid and extensive leaf death at very low population densities. As little as two mites per leaf has be known to kill a to kill a tomato leaflet in one week. The carmine mite is believed to introduce a a potent toxin into the leaf during feeding.
Carmine spider mite infestations in Alberta greenhouses have been rare, but when they have occurred the damage to the plants have been rapid and dramatic and growers have had to resort to chemical control to eradicate the pest.
Aphids
Aphids are not common pests of greenhouse tomatoes. When infestations occur they are usually on plants close to the vents.
The green peach aphid (Myzus persicae) is the most common aphid pest of greenhouse vegetable crops but there are other aphid species that can become a problem in greenhouse tomatoes. These other aphid species include; the melon aphid (Aphis gossypii), the potato aphid (Macrosiphum euphorbiae) and the foxglove aphid (Aulacorthum solani).

Figure 18. The green peach aphid.
Not all aphid biological control agents are equally effective on all aphid species so it is necessary to be sure of the identity of the aphid species in question. All of the species eventually develop winged forms.
Green peach aphids are usually light green in colour, but can be pinkish or yellowish in colour in the fall (Howard et al 1994, Portree 1996). The body is about 1.2 to 2.5 millimetres long and egg shaped. The winged forms can have black or brown coloured heads and black markings on the body.
The melon aphid adults are usually either black or green when there are just a few aphids present, but as the population grows and the aphids become crowded the colours can range from olive green to yellowish green. Melon aphids are about the same size as green peach aphids, 1 to 3 millimetres long, they can be distinguished from the other aphid species by the dark black cornicles and short antennae (Howard et al 1994, Portree 1996).
Potato aphids are quite large, 1.7 to 3.6 millimetres long and the body is wedge-shaped and yellowish green to pink in colour (Howard et al 1994). The head has prominent antennal tubercles that are directed outwards (Howard et al 1994). Potato aphids will drop off the leaves when disturbed (Howard et al 1994).
Foxglove aphids are smaller than potato aphids but larger than melon and green peach aphids. This aphid is a shiny light yellowish green to dark green in colour with a pear-shaped body Howard et al 1994). The only markings on the bodies of wingless adults are darkish patches at the base of the cornicles (Howard et al 1994).
Aphids can be present in the crop very early, even while the plants are just in the seedling stage. They can come in on the transplants as well. Aphids feed by sucking the plant sap. Symptoms of aphid infestation include the development of sticky honeydew on the leaves and fruit. The presence of honeydew on the fruit requires that the fruit be washed prior to going to market.
Sooty mold is often associated with the aphid honeydew, this mold uses the honeydew as a food source and grows to resemble a layer of "soot" on the leaves and fruit. The presence of sooty mold on the fruit also makes washing the fruit a necessity. The growing points, young leaves, flowers and young leaves can be damaged and distorted and in severe infestations flower abortion can occur.
Parasitic wasps provide effective aphid control; Aphidius matricariae for green peach aphid, Aphidius colemani for the melon aphid and green peach aphid and Aphidius ervi for potato aphid. Another parasitic wasp Aphelinus abdominalis is effective against the potato and foxglove aphid. Parasitized aphids become silvery-brown in colour with a small exit hole at the back when the parasite has emerged. The larvae of the midge Aphidoletes aphidimyza feed on most aphid species, but will not feed on gall forming aphids (Don Elliott, pers comm)
Consult your local supplier for information and recommendations on release rates.
Fungus gnats
Fungus gnats are commonly found in practically all greenhouse crops (Howard et al 1994). Fungus gnats are an indicator of moist conditions in the greenhouse and populations generally grow to be quite large early in the year or whenever there is pooling of water on the greenhouse floor.
Adult fungus gnats range from 2 to 3 millimetres in length, while the larvae are 4 to 5 millimetres long. The larvae of the fungus gnats are the damaging stage and feed on the roots. They are generally not a problem in greenhouse tomato and pepper, but can be a serious in cucumbers (Howard et al 1994), especially young plants. Affected plants develop slowly and may eventually collapse if too much of the root system has been damaged (Portree 1996). There is evidence that fungus gnat adults may transport root rot fungi such as Pythium sp. and Fusarium sp. from plant to plant, contributing to the spread of disease caused by these fungi (Howard et al 1994).
Fungus gnats are often confused with shore flies, as both are common in the greenhouse under wet conditions. Shore flies are slightly larger than fungus gnats, and look like scaled-down versions of house flies, while fungus gnats look more like tiny mosquitos that don't bite.
Biological control of fungus gnats is obtained through the use of predatory mites Hypoaspis miles and Hypoaspis aculeifer. Both of these predatory mites also have activity against thrips larvae that move to the base of the plants to pupate. Nematode parasites in the genus Steinernema are applied as a drench to the root zone and kill the fungus gnat larvae by penetrating the larvae and consume them from the inside.
Authored by Dr. James Calpas.